Product Focus- Aseptiquik DC and Aseptiquik STC
Aseptiquik DC Sterile Single Use Connectors
For today’s blog post, we’re going to take a look at one of the product lines we saw great growth in 2016- Colder’s line of Aseptic Connectors, Aseptiquik. Specifically, we’ll look at to of the latest additions to the product line, the Aseptiquik DC and Aseptiquik STC. Both products leverage the versatility of the Aseptiquik product line to bring robust solution to very specific biopharmaceutical applications we’ll examine further in this post.
The first product we want to highlight is the Aseptiquik DC connector. This connector is the first all-in-one single technology to offer both a sterile connection AND disconnection. This allows for quick, easy sterile connections and disconnections outside of a sterile environment or laminar flow hood. No sealers, no welders after transfer is complete, just an intuitive click-pull twist. The disconnect is even valved, eliminating the need for a pinch clamp.
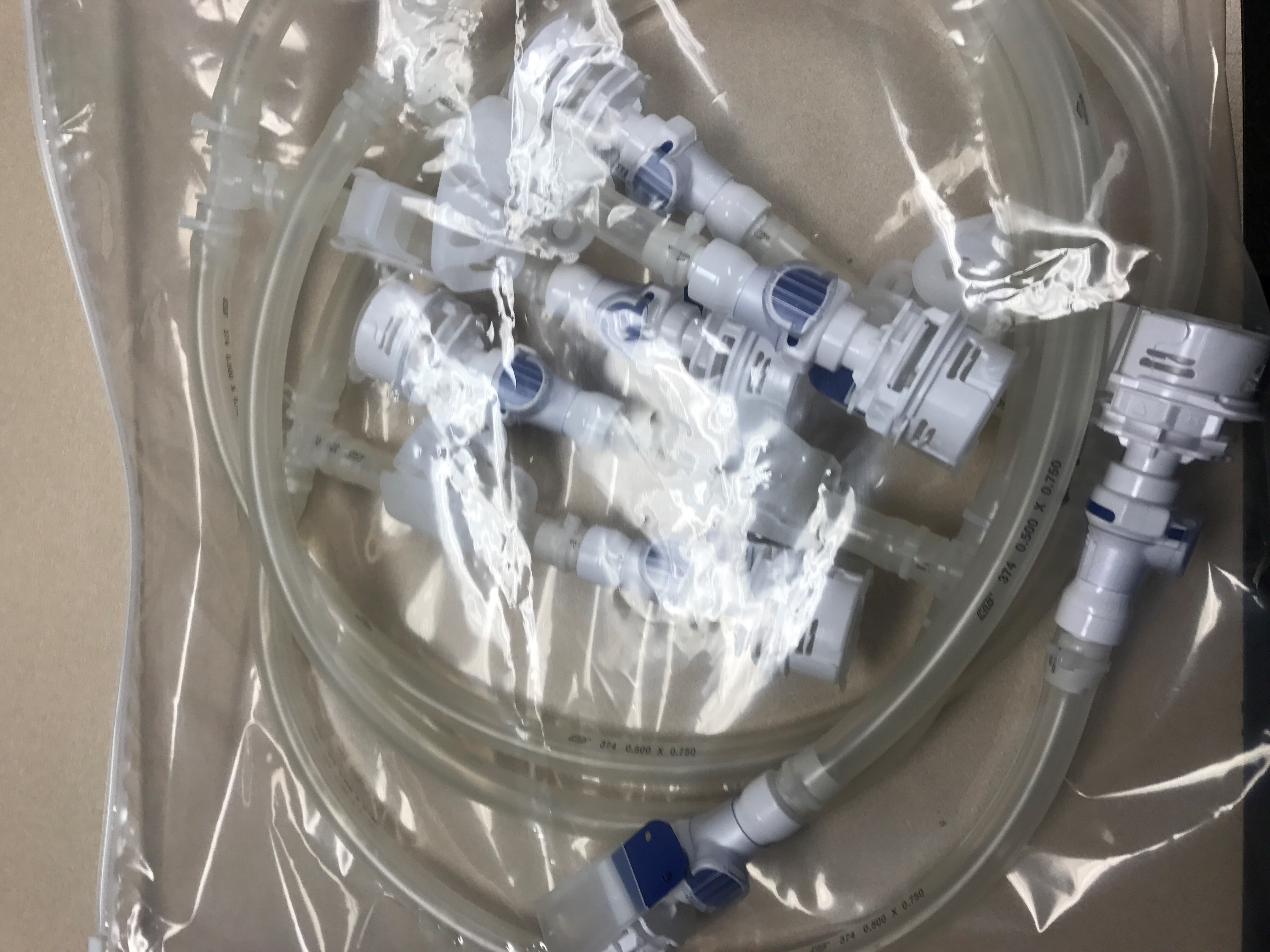
Where is this applicable, you may ask? Let’s look at a recent application we helped a customer with. A contract manufacturer recently approached us to prototype a disposable manifold for them. This client had an application where they were recirculating buffer media over the course of a campaign and over the course of the campaign needed to dose into a disposable bioreactor already equipped with Colder Aseptiquik connectors. Because the campaign ran over a period of weeks, multiple ports were required off the manifold. After buffer was transferred into the bioreactor, a sterile disconnect was preferred. So with the help up Holland, the customer was able to implement a five port single use manifold equipped with seven Aseptiquik DC connectors, allowing for a sterile connection to the plastic media bag and (5) separate sterile connections to the bioreactor.

Single Use 5 Port Manifold with Colder Aseptiquik Connectors
The second product we wanted to highlight is the Aseptiquik STC. Available with both gendered and gender less sterile connectors, the Aseptiquik STC incorporates Colder’s Steam Thru II connector. This combination gives biopharmaceutical manufacturers the flexibility to marry single use and stainless technologies.
Why would this be useful? Imagine you have a stainless steel buffer and bulk product or media received in disposable bag. You need to find a way to steam the vessel and maintain the sterile barrier when you connect the media bag. As we’ve highlighted in a previous post, during an SIP cycle, a Steam Thru II connector is in the “steam on” position, allowing steam to flow through the process equipment, through the Steam Thru connector to a trap, creating a sterile connection between the connector and the stainless vessel. The Aseptiquik connector is then connected to a single use piece of equipment with a matching sterile connector. After this connection is made, the thumb latch on the Steam Thru II connector is pushed and the valve is locked into the fluid flow position, allowing for sterile transfer in or our of the stainless vessel, to or from a single use bag. Because we use a Steam Thru II connector, once transfer is complete, the Steam Thru II can be re-engaged and allow for a “steam off”.
In sum, as biopharmaceutical manufacturing becomes increasingly complex, manufacturer next generation therapies, it is critical that our technologies continue to evolve. The Aseptiquik STC and Aseptiquik DC are two new products we at Holland have recently applied to help customers to address difficult process challenges.
If you have a specific question about your biopharmaceutical processing application, contact a Holland Sales Engineer today.








